 
It is a nonmetal with properties that are intermediate between sulfur and tellurium. Selenium is the third element in group 16 and is less reactive than oxygen but more reactive than tellurium. However, tellurium is not as readily reactive as oxygen or sulfur. It is a brittle, crystalline solid that can react with oxygen to form tellurium dioxide. Tellurium is the fourth element in group 16 and is less reactive than both oxygen and sulfur. It requires higher temperatures or the presence of a catalyst to undergo reactions. 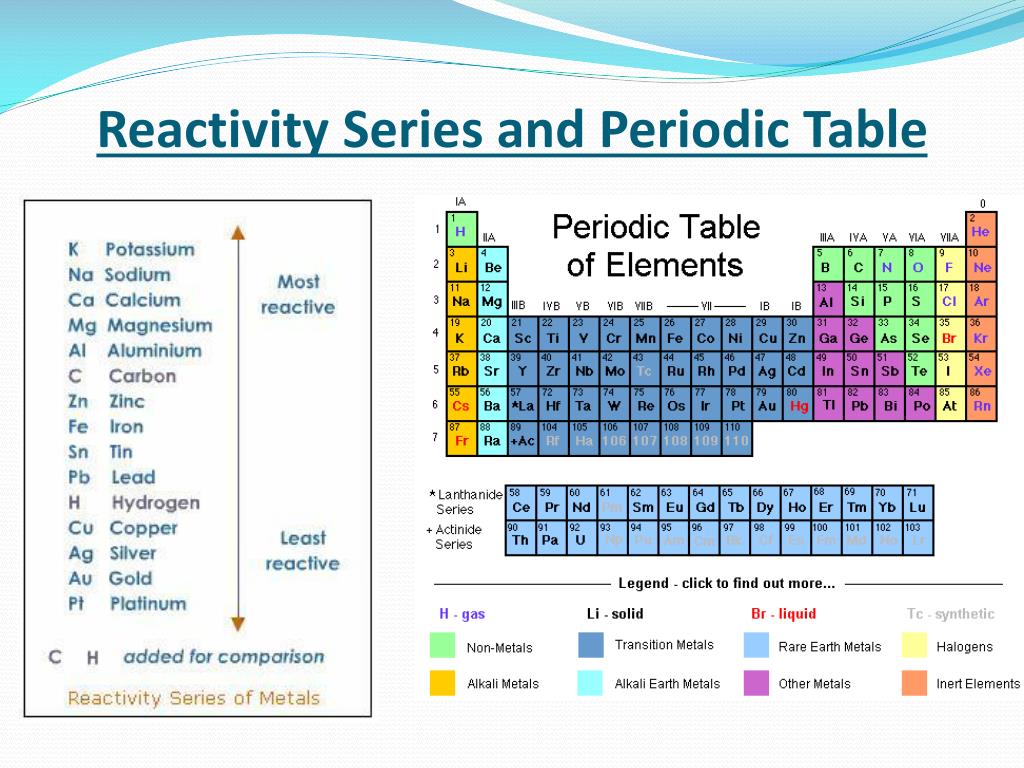
However, compared to oxygen, sulfur is less reactive. It is a yellow solid that can react with oxygen to form sulfur dioxide. 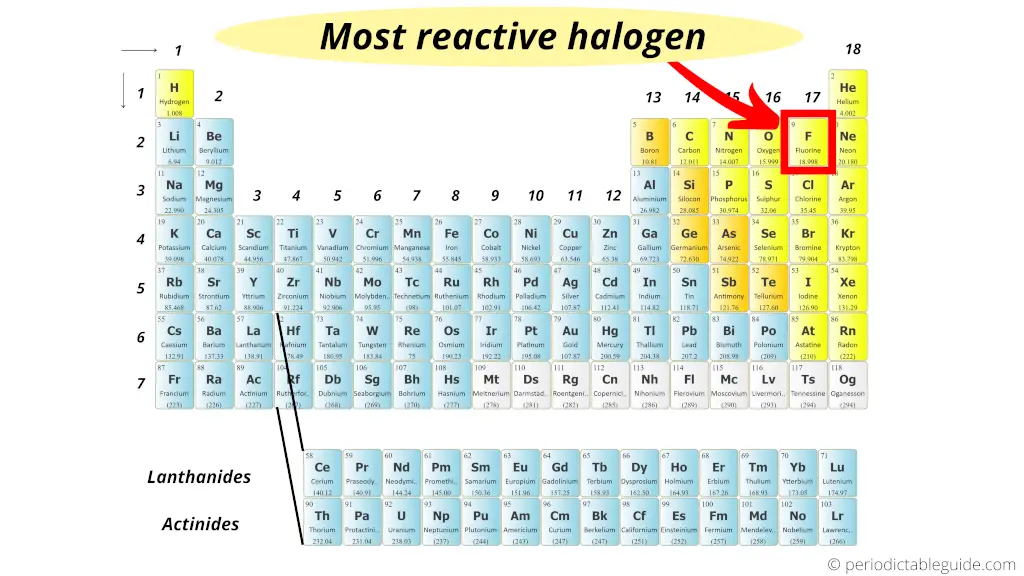
Sulfur is the second element in group 16 and is also quite reactive. Oxygen is essential for supporting combustion, and many substances burn in the presence of oxygen. It is a highly reactive nonmetal that readily forms compounds with other elements. Oxygen is the first element in group 16 and is known for its high reactivity. As a result, the reactivity of group 16 elements increases from top to bottom. This is because the outermost energy level of the atoms gets farther away from the nucleus, making it easier for the atoms to gain or share electrons. Reactivity generally increases as you move down a group in the periodic table. Group 16 elements are also commonly referred to as the oxygen group or the group of oxygen family. Oxygen belongs to group 16, also known as the chalcogens, which consists of oxygen, sulfur, selenium, tellurium, and polonium. The most reactive element of group 16 is oxygen. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
